Semaglutide is the generic name of a medication that falls under the category of glucagon-like peptide-1 receptor (GLP-1) agonist. Semaglutide is approved by the FDA for use in the United States under 3 different brand names: Ozempic, Rybelsus, and Wegovy. In this article, we will clarify the differences between the 3 brands of semaglutide and discuss the role of semaglutide as a weight loss medication, including how it works, who is a candidate, safety, side effects, and how to best obtain the medication.
Ozempic vs Rybelsus vs Wegovy
Before discussing how semaglutide works, let’s resolve any confusion surrounding the different brand names and approved indications. Semaglutide was first approved by the FDA in December 2017 under the brand name Ozempic as a once weekly injection for the treatment of type 2 diabetes mellitus. Ozempic comes in the following doses: 0.25, 0.5, 1.0, and 2.0 milligrams. Ozempic is administered once-weekly via a pen that has a dose dialer on one end and an attachment for a pen needle at the other.

An oral version of semaglutide taken once daily was created by Novo Nordisk and released into the market in 2019 under the brand name Rybelsus. Like Ozempic, Rybelsus is FDA approved for the treatment of type 2 diabetes mellitus. Rybelsus comes in the following doses: 3, 7, and 14 milligrams. It is taken as one tablet a day. Rybelsus must be taken at least 30 minutes before the first food, drink, or medication of the day, with no more than 4 ounces of plain water1.
In March 2021, a pivotal clinical trial called STEP 1 was published showing once-weekly injectable semaglutide resulting in an impressive 14.9% average reduction in body weight in individuals with obesity without diabetes mellitus. This led to the FDA’s approval in June 2021 of a higher maximum dose once-weekly injectable semaglutide under the brand name Wegovy. Wegovy is an anti-obesity medication (AOM) approved “as an adjunct to a reduced calorie diet and increased physical activity for chronic weight management2.” Wegovy comes in the following doses: 0.25, 0.5, 1.0, 1.7 and 2.4 milligrams. Wegovy is administered once-weekly via an auto-injector pen. There is no dose dialing or pen needle attachment like the Ozempic pen.
In summary, all 3 are the same drug (semaglutide). The differences are the approved indications (diabetes mellitus vs weight loss) and route of administration (subcutaneous injection vs oral).
How Do Ozempic and Wegovy (semaglutide) Work for Weight Loss?
Semaglutide is a glucagon-like peptide-1 receptor (GLP-1) agonist. It works by enhancing the effect of the naturally-occurring hormone GLP-1. GLP-1 is produced by the intestine and acts on different parts of the body:
– It promotes insulin production and release by the beta-cells of the pancreas.
– It slows the emptying of food contents from the stomach.
– It promotes the feeling of fullness (satiety) by acting on the brain, particularly at the level of the hypothalamus.
The weight loss effect is believed to be due to increased satiety and fullness, ultimately reducing caloric intake.
How Much Weight Can You Lose on Wegovy?
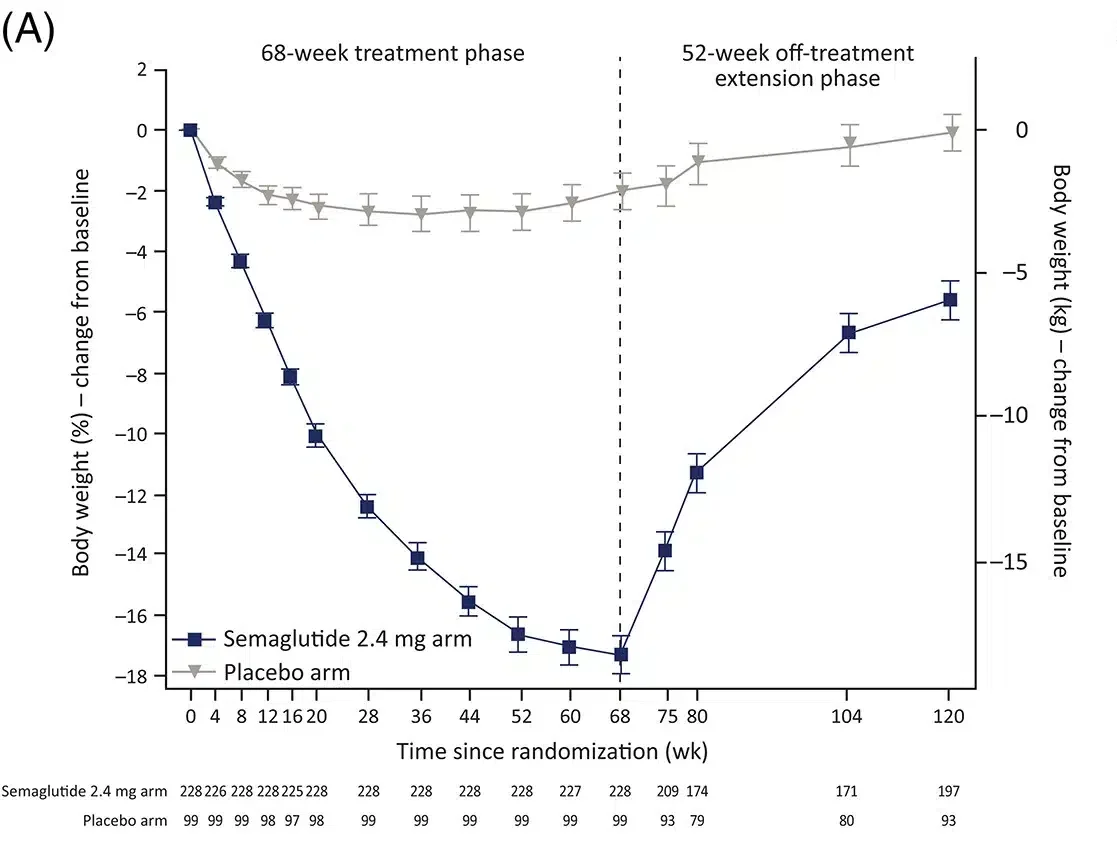
Let’s look at the STEP 1 trial to accurately answer this question. This trial enrolled 1,961 individuals with BMI ≥ 30 or BMI ≥ 27 + 1 weight-related condition who did not have diabetes mellitus. These individuals were randomly assigned to once-weekly injectable semaglutide at a maximum dose of 2.4mg (Wegovy) or placebo, plus lifestyle intervention. At the end of 68 weeks, individuals in the semaglutide group lost an average of 14.9% of their body weight, compared with 2.4% in the control group (see below).
A 14.9% reduction in body weight has the potential to improve, prevent, or possibly reverse certain disease processes. In individuals with prediabetes, 7% – 10% body weight loss is effective for preventing progression to diabetes3. 5% – 10% body weight loss will decrease the liver’s fat content in nonalcoholic fatty liver disease4.
Do you gain weight back after stopping Wegovy?
The STEP 1 trial extension has excellent data shown above to answer this question. At the end of the 68-week STEP 1 trial, treatments were discontinued (including lifestyle intervention) for all participants. 333 participants entered into the extension trial and were followed for an additional 52 weeks off treatment. Of the 333 participants, 232 were previously in the semaglutide arm and 101 in the placebo arm. For reference, the 232 participants in the semaglutide arm achieved an average body weight loss of 17.3%, and the 101 participants in the placebo arm achieved an average body weight loss of 2.0%.
At the end of the 52-week extension, previous semaglutide participants regained 11.6% of lost body weight and placebo participants regained 1.9% of lost body weight. The net result was a loss of 5.6% of body weight in semaglutide participants and 0.1% in placebo participants.
At the end of the total 120 weeks, we see a regain of two-thirds of body weight and bodyweight seems to be leveling off. However, we are unable to conclude whether this is where the regain ends or if it continues beyond the study’s time frame.
This study demonstrates the chronic nature of obesity as a disease and the need for long-term treatment to improve and maintain body weight and health.
Who is a Candidate for Wegovy or Ozempic?
Wegovy is an anti-obesity medication (AOM) indicated “as an adjunct to a reduced calorie diet and increased physical activity for chronic weight management2” in adults meeting the following criteria:
- Body Mass Index (BMI) ≥ 30kg/m2
- BMI ≥ 27kg/m2 with the presence of a weight-related condition, such as high blood pressure, prediabetes, type 2 diabetes mellitus, elevated cholesterol, heart disease, obstructive sleep apnea, nonalcoholic fatty liver disease (NAFLD), etc.
Ozempic is approved for the treatment of type 2 diabetes mellitus. You may be a candidate for Ozempic to improve blood sugar control in the management of type 2 diabetes mellitus. Ozempic may be used off-label for weight loss, without the presence of type 2 diabetes mellitus, at the discretion of the managing physician. In such a situation, the BMI criteria above would apply.|
Semaglutide Safety and Side Effects for Weight Loss
Semaglutide, both in the form of Wegovy and Ozempic, is contraindicated in individuals with a personal or family history of medullary thyroid cancer and in individuals with Multiple Endocrine Neoplasia (MEN) syndrome type 2.
Common side effects with semaglutide include nausea, vomiting, diarrhea, abdominal pain, constipation, and headaches. Strategies for reducing or managing side effects include a slow titration, adequate hydration, and use of medication for symptom control as needed.
Less common but serious adverse effects seen in the clinical trials are pancreatitis (inflammation of the pancreas), gallbladder disease (such as gallstones), and worsening of diabetic retinopathy.
Where Can I Get Wegovy or Ozempic for Weight Loss?
Wegovy or Ozempic is best obtained through a board-certified obesity medicine physician. An obesity medicine physician can evaluate your medical history and create a comprehensive, personalized plan that includes nutrition, physical activity, and the best weight loss medication for you. Dr. Aleem Kanji is a board-certified obesity medicine specialist and endocrinologist who can evaluate your candidacy for Wegovy or Ozempic and create a personalized weight loss plan for you.